 The change in the internal energy of the system, U. W is positive when more work is done by the system than on it. We will assume the expansion process is quasi-equilibrium, and the piston moves up an infinitesimal distance d. W is the total work done on or by the system. Upon receiving heat, the gas will tend to expand, pushing the piston up. The gas in the cylinder exerts an upward force, F=PA, where P is the gas pressure, and A is the cross-sectional area of the piston. For example, the system could be a sample of gas inside a cylinder with a movable piston, an entire steam engine, a marathon runner, the planet Earth, a neutron star, a black hole, or even the entire universe. The change in the volume of the cylinder ( V) as the piston moves a distance d is V A h, as shown in Figure 18.1.4. Let us consider a piston-cylinder device, as illustrated in Figure 4.3.2. The application of thermodynamic principles begins by defining a system that is in some sense distinct from its surroundings. Alternatively, if the external pressure is greater than Pint (b), the gas will be compressed, and the surroundings will perform work on the system. 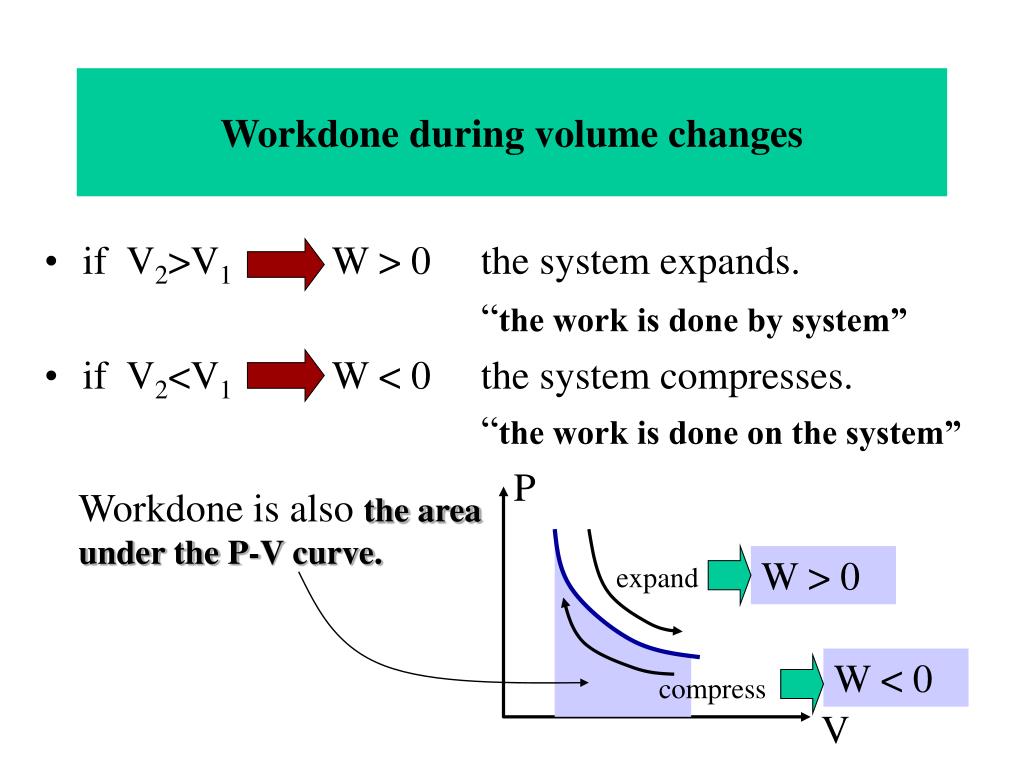 
Work done by gases is also sometimes called pressure-volume or PV work. In thermodynamics, an adiabatic process (Greek: adibatos. Gases can do work through expansion or compression against a constant external pressure. Work associated with the expansion and compression of a gas is commonly called boundary work because it is done at the boundary between a system and its surroundings. When an ideal gas is compressed adiabatically work is done on it and its temperature. Figure 4.3.1 Work done due to a force acting on a block over a distance 4.3.1 Boundary work 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
